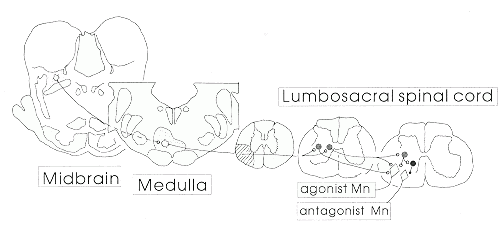
 |
| Figure 1 Schematic drawing of brainstem and spinal cord neuronal
systems involved in the control of locomotion. |
It is now widely accepted that central nervous system (CNS) neurons have the potential for regeneration (see Cheng in this issue, pages 87-89), but the goal of restoration of function through these means will be achieved only after considerable progress is made in methods of stimulating the regenerative capacity of specific populations of neurons. If efforts to produce regeneration of spinal cord pathways are to be successful in restoring locomotion, it is the reticulospinal locomotor command neurons which must be made to grow and form synapses with their appropriate targets. This requires that these cells be successfully identified and characterized, and the factors determining their growth and synapse formation be determined. It is likely that these factors will include target-specific growth factors that promote the survival and growth of this particular cell type, extracellular matrix molecules which may have a role in axonal guidance, and genes within the cells which must be expressed in order for them to respond to the signals which originally determined the formation of functional connections. Factors such as these are likely to be unique for specific functional systems, and the same rules which allowed the cells to grow to the right places and make the right connections during development may be reapplied, with help from exogenous influences provided by clinicians, to achieve functional regeneration.
Numerous efforts are underway around the world to use transplantation methods for restoring lost spinal cord function, and this research also depends upon the successful localization and identification of neurons involved in the functional system which is to be replaced. This approach is similar to that using adrenal or fetal brain cells transplanted into the basal ganglia of Parkinson's disease patients. In order to develop transplantation methods which have any hope of success for restoring the command for initiation of locomotion, it is necessary to identify the appropriate cells for transplantation and the appropriate sites for placement of the transplanted cells.
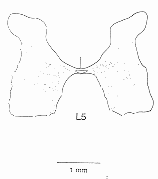 |
| Figure 2 Locations of cells expressing c-fos in the lumbosacral enlargement. All labelled cells found within 17 sections from the L5 segment are illustrated. The expression of c-fos was induced by a brainstem stimulus which gave rise to locomotor activity |
The characteristics and patterns of activity during locomotion of cells in the regions with c-fos expression have also been determined using in vivo electrophysiological methods. One such cell, stained intracellularly with a fluorescent dye, is illustrated on the front cover of this issue. Cells which are excited by stimulation of the MLR have been located in the medial part of the gray matter, near the central canal, and the pharmacology of neurons in this region has been examined in spinal cord slice preparations by our collaborators at the University of Toronto (J MacDonald, S Hochman). Figure 3 illustrates results from one such cell, which was made rhythmically active by application of an excitatory amino acid which we recently demonstrated could initiate locomotion when applied directly onto the spinal cord in adult animals.2 The rhythmic activity observed in the spinal cord slice persists when the action potentials within the slice are blocked with tetrodotoxin. This proves that the capacity for rhythmicity is intrinsic to these cells, and does not depend upon the interconnections in a network of spinal interneurons. This work, which was done as a part of the Network of Centres of Excellence program, provides the first demonstration of pacemaker-like properties of locomotor neurons in the mammalian spinal cord.
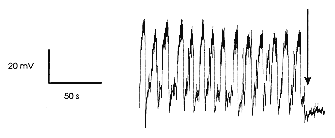 |
| Figure 3 Whole-cell patch clamp recording of membrane potential
oscillations produced by local application of NMDA (N-methyl-D-aspartate)
(20 |
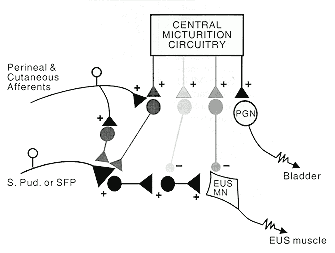 |
| Figure 4 Schematic drawing representing the spinal organization for the control of parasympathetic preganglionic motoneurons (PGN) to the bladder, and motoneurons to the external urethral sphincter (EUS). + Excitatory synapses; - lnhibitory synapses; Mn Motoneuron; SFP Superficial perineal neuron; SPud Sensory pudendal neuron |
The ability to investigate neurons involved in locomotion in vitro is expected to provide sufficient details regarding the properties of these cells to allow successful pharmacological manipulation of their activity. This basic understanding of discrete neuronal populations which contribute to the initiation of locomotion should lead eventually to new drug treatment strategies for patients with locomotor impairment, much as a basic understanding of presynaptic inhibition and the pharmacology of the cells involved has led to the development of the intrathecal baclofen treatment for spasticity (see Nance et al, pages 90-91).
Another spinal cord functional system of considerable clinical and economic importance is the system for control of micturition. Figure 4 illustrates the recent progress on the neural control of external urethral sphincter function which has taken place in Susan Shefchyk's laboratory within the Spinal Cord Research Centre. Shefchyk3 was the first to demonstrate that the CNS, under conditions where it is deprived of sensory feedback, contains the central micturition circuitry necessary for coordination of bladder contraction with external urethral sphincter relaxation. She and her collaborators have gone on to demonstrate that sphincter relaxation depends on the activity of a group of spinal inhibitory interneurons4 and have localized the excitatory interneurons with input from peripheral sensory pathways5,6 which could contribute to bladder-sphincter dyssynergia after CNS injury or disease. They have also provided new evidence that there is presynaptic control of sensory afferents to sphincter motor neurons.7 This finding can explain the efficacy of baclofen for reduction of dyssynergia, because baclofen acts as an agonist for the transmitter normally released at presynaptic inhibitory sites.
Progress in the identification and characterization of discrete populations of cells of functional importance will continue to be one of the major goals of the Spinal Cord Research Centre. In the coming years, the centre plans to continue research into this, coupled with clinical studies which involve the development of new methods of applying the basic knowledge as it accumulates, and regeneration studies relating to the growth and synapse formation of the relevant neuronal populations.
2. Douglas JR Dai X Noga BR Jordan LM Intrathecally
administered excitatory amino acid agonists and antagonists demonstrate
that NMDA receptor activation is necessary but not sufficient for the initiation
of locomotion in the adult cat J Neurosci (in press) ![]()
3. Shefchyk SJ Effects of lumbosacral deafferentation
on pontine micturition centre evoked micturition in the cat Neurosci Lett
1989 99 175-80 ![]()
4. Fedirchuk B Shefchyk SJ Membrane potential changes
in sphincter motoneurons during micturition in the decerebrate cat J Neurosci
1993 13 990-1000 ![]()
5. Fedirchuk B Hochman S Shefchyk SJ An intracellular
study of perineal and hindlimb afferent inputs onto sphincter motoneurons
in the decerebrate cat Exp Brain Res 1992 89 511-6 ![]()
6. Fedirchuk B Song L Downie J Shefchyk SJ Spinal
distribution of extracellular field potentials generated by electrical
stimulation of pudendal and perineal afferent in the cat Exp Brain Res
1992 89 517-20 ![]()
7. Angel MJ McCrea DA Fyda D Shefchyk SJ PAD of perineal
afferents evoked by stimulation of sensory afferents and during micturition
in the cat Soc Neurosci Abst 1992 18 (217) 19 ![]()
8. Noga BR Kettler n Jordan LM Locomotion induced
in mesencephalic cats by injections of putative transmitter substances
and antagonists into the medial reticular formation and the ponto-medullary
locomotor strip J Neurosci 1988 8 2074-86 ![]()
9. Noga BR Kriellaars DJ Jordan LM The effect of
selective brainstem or spinal cord lesions on treadmill locomotion evoked
by stimulation of the mesencephalic or pontomedullary locomotor regions
J Neurosci 1991 11 1691-700 ![]()
10. Jordan LM Brownstone RM Noga BR Control of functional
systems in the brainstem and spinal cord Curr Opin Neurobiol 1992 2 794-801 ![]()